Catalytic Alkynylation of Ketones and Aldehydes Using Quaternary Ammonium Hydroxide Base | The Journal of Organic Chemistry
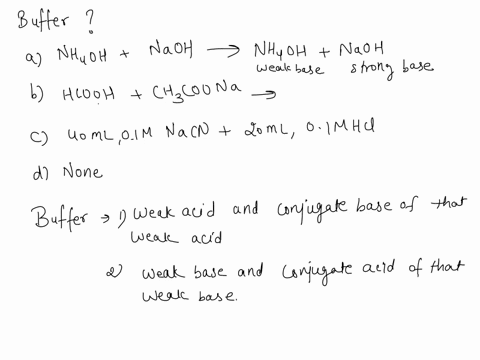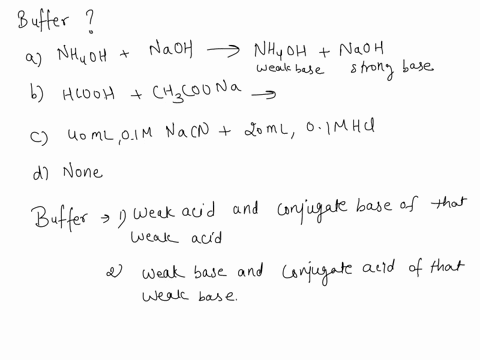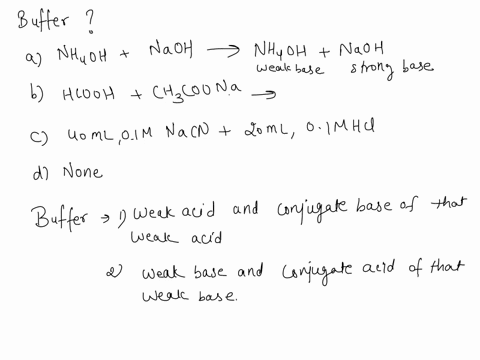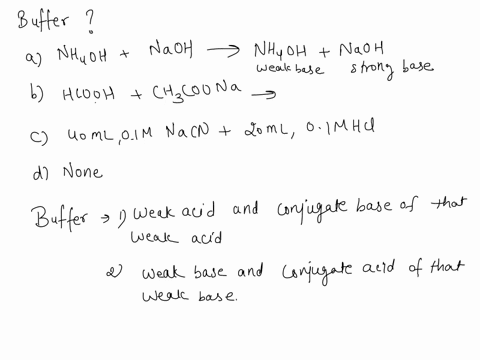
SOLVED: Which is a buffer solution? Select one: a. NH4OH + CH3COONH4 b. K2SO4 + H2SO4 c. NaOH + CH3COONa d. NaOH + Na2SO4
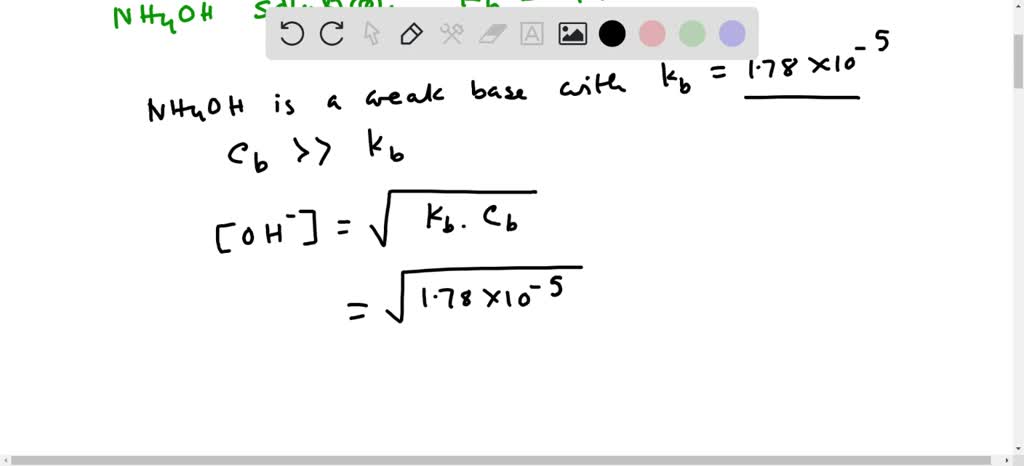
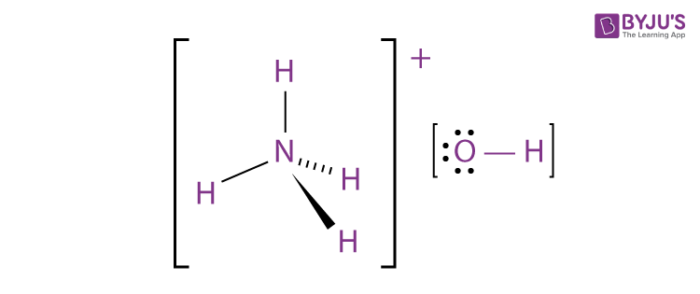
SOLVED: Answer the following equation: Ammonium Hydroxide, NH4OH, is a weak base. Calculate the pH of a 0.60 M solution of ammonium hydroxide. Kb = 1.78 x 10^-5.

Reaction of 1 with NH4OH in DMSO. (a) UV-vis spectral observations in... | Download Scientific Diagram










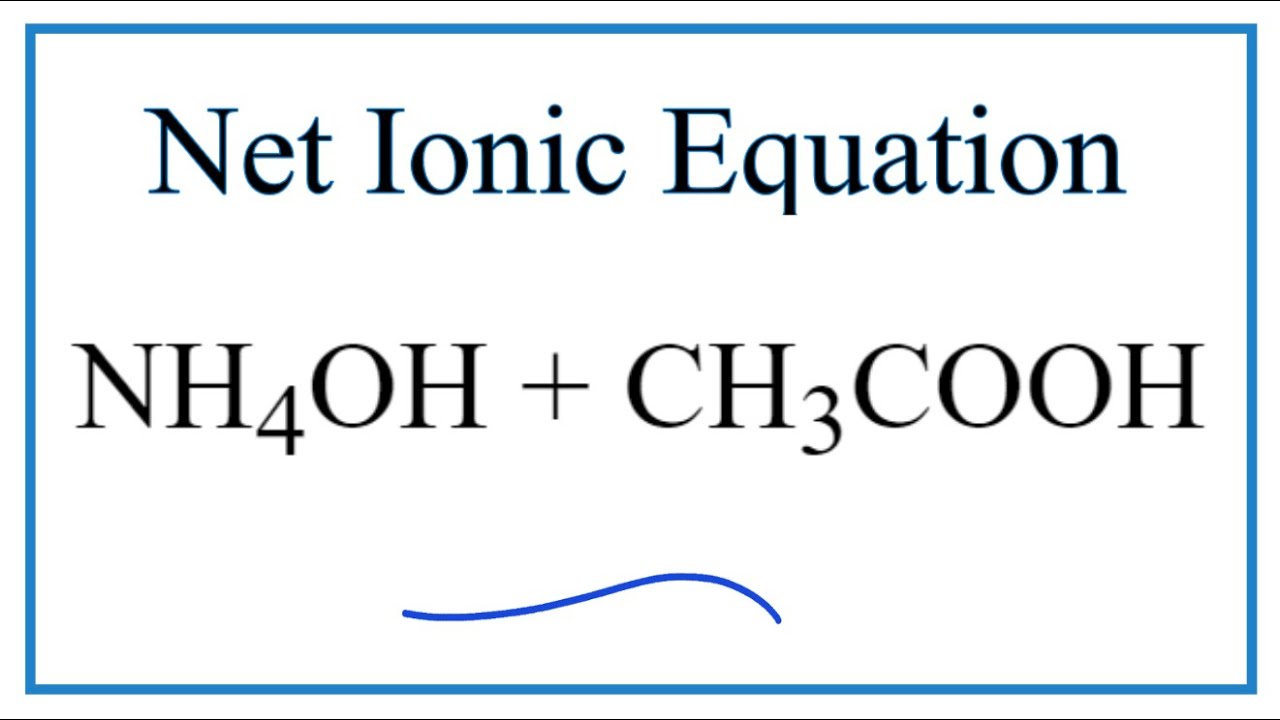
![ANSWERED] When aqueous solutions of NH4OH(aq) and Cu... - Organic Chemistry ANSWERED] When aqueous solutions of NH4OH(aq) and Cu... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/52914656-1658983365.4892888.jpeg)