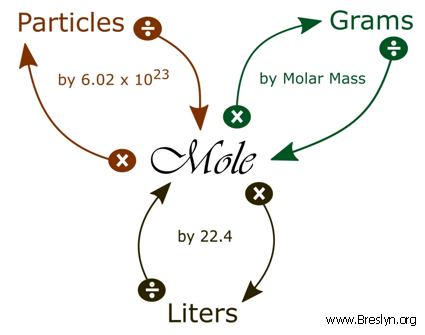
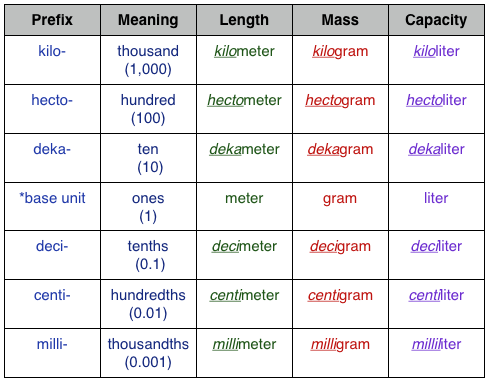
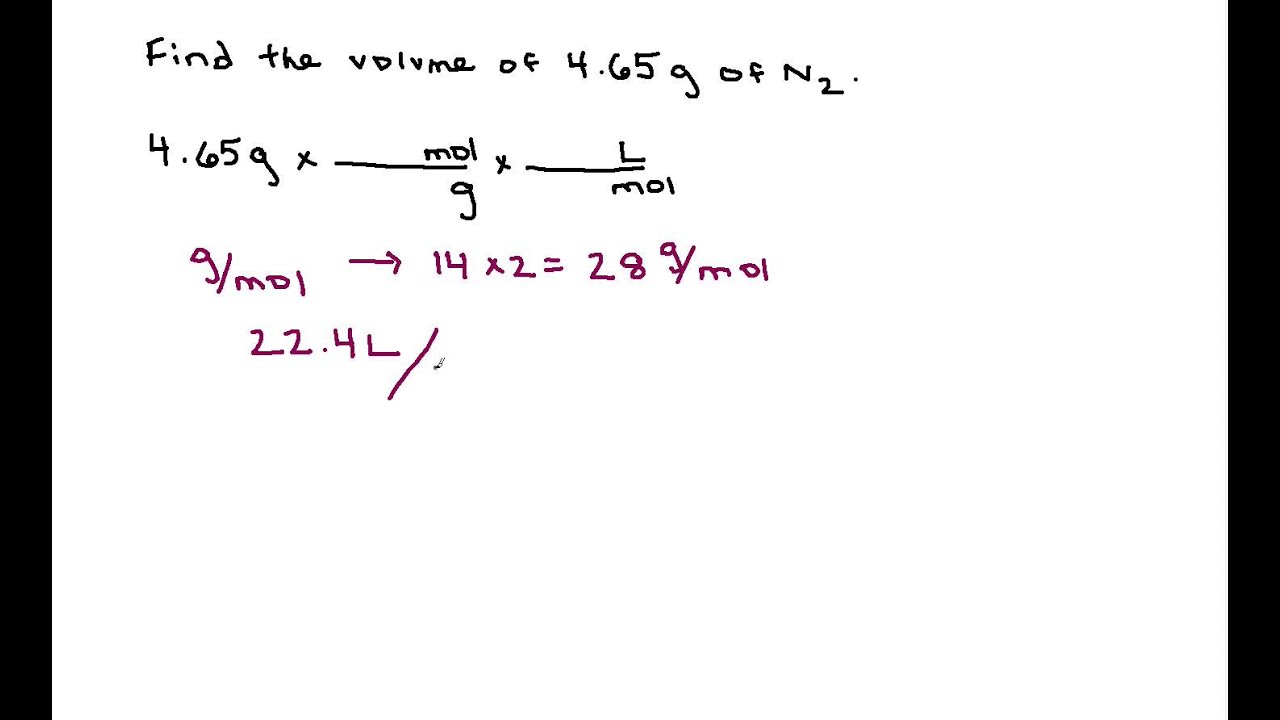SOLVED: How many grams of oxygen (O2) would there be in 1.00 liter of oxygen at STP? What volume, expressed in liters, would 50.0 grams of fluorine (F2) occupy at STP?

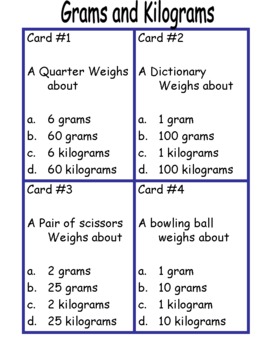
SOLVED: 1. How many grams of Cl2 gas are in a 5.00-liter tank at STP? 2. The pH of 0.01 M LiOH is 3. At STP, what is the volume of 7.00
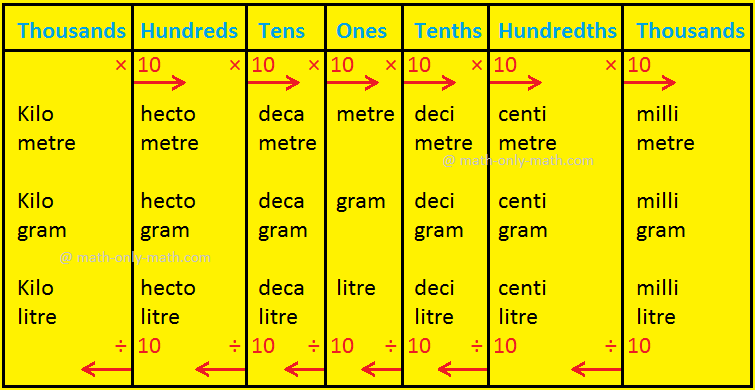
Tde is 15. Compute the mass in grams of KCIO3 necessary to produce 67.2 litres of oxygen at S.T.P. according to the reaction 2KCIO3 (s)2KCI (s) + 302 (g) [Molecular weight of